Stranger Genes: How Seattle Scientists Are Advancing Gene Editing
Image courtesy the Allen Institute for Cell Science
The day begins with an empty glass, filled component by component—a packet of probiotics; scoops of glutamine, calcium magnesium citrate, and B complex; drops of Vitamin D; fruit juice—until it has all the appeal of a giant pill. Four over-easy eggs float in homemade chicken broth, sprinkled liberally with salt. Breakfast. She eats it standing up at the kitchen counter. Eating and drinking are chores for Julie Negrin, not pleasures.
In her downstairs bathroom, where colored Post-It notes cover her vanity mirror and bottles of medication litter the sink, she takes her dose of hydrocortisone. She’ll return to the bathroom three times in the span of an hour. But not to take her medicine.
Negrin, 46, grew up in Redmond, and most of that growing up she spent enduring bowel trouble, thanks to Crohn’s disease, which she inherited on her mom’s side. On top of that, as an adult, she battled melanoma cancer. In December 2013, while she was living in San Diego, working as a nutritionist, things got worse. Negrin discovered she had colon cancer. “I’m just a kind of organism,” she says, “that in any other era wouldn’t have lasted this long.”
That diagnosis led to another: Lynch syndrome, a rare genetic disease from her father’s side that predisposes her to a variety of cancers. A PET scan at that time also lit up her ovaries, but she postponed her first, then second follow-up appointment. When she finally went in, she asked a friend to come. It was cancer. Again. “I knew if there was cancer in my ovaries,” she says, “it could be everywhere.”

Julie Negrin hopes new gene-editing technology will save the lives of others with Lynch syndrome, including possibly her nieces and nephews.
Image: Ian Bates
People with Lynch syndrome have an 80 percent chance they might develop colon cancer; women with the disease have a 71 percent chance of developing endometrial cancer (in the uterus), per the American Association for Cancer Research.
Sure enough, in March 2014, when Negrin woke up after surgery to remove most of her colon, ovaries, and—as a preventative measure—her uterus, doctors said they found a third cancer.
She stopped working. Moved into her parents' house on Mercer Island. Staying alive became a full-time job.
She lost her hair during chemotherapy, and when it grew back she styled it into a mohawk, which she now wears with red frosted tips. She is cancer free, but she doesn’t know how long it’ll last. The numerous surgeries prompted other problems with her GI tract and devastated her body, and she suffers from chronic dehydration. At night, she stretches, stimulates GI tract muscle movement through massages, and intakes more IV fluids and estrogen, progesterone, and testosterone.
When she’s low on testosterone, she can’t get out of bed. Her old chest port became infected last November, leaving her with a large scar by her collarbone.
A doctor once told Negrin: “We are taught to look for horses in medicine,” she recalls. “You’re the zebra. You’re the case we get on the medical board.”
There’s nothing out there Negrin hasn’t already tried. Chances are she’ll battle her bowel problems and cancer predispositions for the rest of her life. But a few years ago, she found something that gave her hope—she began reading stories about a new gene-editing technology that could, someday in the future, help people like her. An enzyme named CRISPR-Cas9 attaches itself to targeted parts of a cell's DNA—making it possible to pinpoint genes that are directly responsible for developing diseases and cut that disease out of the DNA sequence.
Scientists are still perfecting this tool. And some of them are hard at work just miles away from where Negrin stands, motoring through the daily routine of keeping herself alive.
At the corner of Mercer and Westlake in South Lake Union, the hub of Seattle’s seemingly unstoppable tech boom—where Amazon bumps against cutting edge medical science from the likes of the Fred Hutchison Cancer Research Center—stands a 270,000-square-foot, $100 million building shrouded in glass: the Allen Institute. Founded by Microsoft cofounder and Seahawks owner Paul Allen, the institute privately funds research in brain and cell science largely through Allen’s own investment of more than $1 billion.
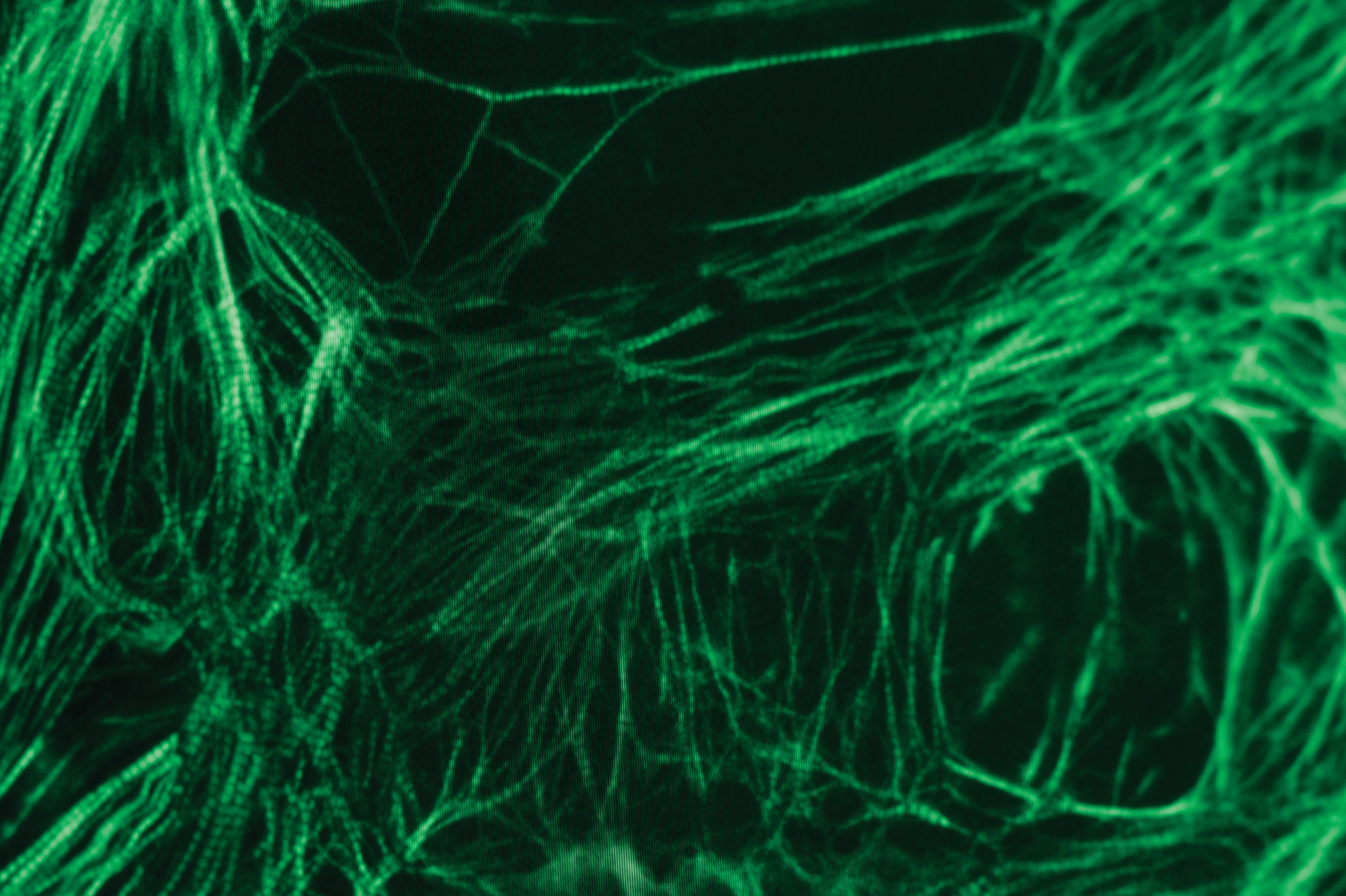
Past the front desk cam that snaps your photo for security clearance and the Allen Institute promotional video that twitches on a TV monitor encompassing nearly an entire wall and up five floors is the cell lab. There, on a recent sunny day, a team of researchers peered through the microscope at a cluster of 10 million healthy adult stem cells. Once skin cells, they had been reprogrammed to become stem cells and shipped to Seattle, where these scientists could perform their magic. In the natural light that streamed through the lab, the researchers methodically tagged cell organelles with a protein derived from a jellyfish most commonly found in the Puget Sound. Ten years ago, a Japanese chemist won the Nobel Prize for discovering the jellyfish’s green fluorescent protein can be used to tag parts of a cell.

Allen Institute researchers collect cells and place them in petri dishes to be zapped via the CRISPR-Cas9 method.
Image: Ian Bates
After she measured out just the right amount of cells with a handheld tool known as a pipette, research associate Maggie Fuqua transferred about 800,000 of them (just 100 microliters) from the small tube to a machine to zap the cells with a jolt of electricity and briefly open up the cell membranes’ pores to insert enzymes, the only sign of that jolt a little burst of bubbles emitting from the bottom of the cells’ container.
A computer then displayed the image of a dish after it received the jolt: heart cells, small circles of nuclei with a pulse that rippled across the gray screen. Placed in the dark, exposed to ultraviolet light, and boom—in a flash, lime green color illuminated strands of tissue, a network of spider webs within the cells on a screen. It was like staring at another world, the Upside Down, say, from Stranger Things. As the cells pulsed, the ropes of tissue stretched out and tightened in waves, then reverted back to slackened straight lines.
That tagging, that lighting of precise sites in a cell, could lead to cures we once thought impossible. That tagging could someday lead to changing the unchangeable—DNA sequence. What once had been thought possible only in science fiction would soon become reality.
This is the CRISPR-Cas9 method, the one Julie Negrin had read about, and here’s how it works. It takes a cell and searches inside it for a specific location and uses three components, the CRISPR molecule, the Cas9 enzyme, and a “template” DNA. The CRISPR molecule is essentially a messenger—it tells the Cas9 enzyme where to go. Cas9 then attaches itself to that location and slices that part of the gene. Cuts that cell portion up, and replaces it with the template DNA, which is fused with bioluminescence that lights up under a fluorescent microscope.

After cells are edited, they're prepared in frozen vials and delivered to labs all around the world.
Image: Ian Bates
Theoretically, once you find the part of the mutant cell that causes the disease, and slice it out of the gene, you're essentially deleting the disease out of the DNA sequence.
The ultimate dream, then, is to course correct genes for people who have truly debilitating conditions—cancer, autoimmune diseases, birth defects.
Are we there yet? No. But we’re probably not as far off as you might expect.
Researchers around the world, using these tagged CRISPR-edited cells, are conducting clinical trials for an array of conditions including sickle cell anemia, certain cancers, and HIV. One gene therapy that’s produced promising results so far could be close to correcting blindness by fixing eye pigments; several other scientists are working on regenerating muscle for muscular dystrophy. The possibilities are seemingly endless.

Allen Institute for Cell Science executive director Rick Horwitz says DNA editing could be the dream of sci-fi finally realized.
Image: Ian Bates
That’s been medicine’s goal from day one, says Allen Institute for Cell Science executive director Rick Horwitz. “This technology offers the potential to have a huge impact on human health.”
The institute doesn’t produce specific research on diseases; it provides the cells that are used for that research. And unlike other companies that charge labs tens of thousands of dollars to access those tagged cells, the institute's mission is to create open-source science on an industrial scale. These cells are readily available for any kind of research; and just in the past two years, the Allen Institute has processed 250 orders of cell lines for labs located in eight different countries.
Understand the cells, and every player within them, and you understand how the disease can develop in the body and how to repair it. If there’s someone with heart disease, for example, you could identify the problem in how the strands are interacting within the cells.
And because of CRISPR-Cas9, we can see it now. Without it, it’s like conducting surgery blind.
“Imagine that you want to understand how a car works,” Maggie Fuqua says, of medical science before CRISPR technology, “but you open the hood, and everything is invisible.”
A saltwater mini reef, set in an aquarium tank, sits in Beno Freedman’s University of Washington School of Medicine lab office, where he uses the Allen Institute’s crafted cells for his own research. As kids growing up in Chicago, Freedman and his twin brother packed their household with pets—Freedman sustained an obsession with aquariums, including a 90-gallon tank of fish and corals, while his twin kept lizards, newts, and salamanders. The reptiles’ ability to regenerate limbs fascinated the young Beno.
That inspired his first undergraduate project at the University of Pennsylvania, which focused on regrowing body parts. This fascination shifted to regenerating organs when his uncle Barry in St. Louis needed a second kidney transplant after his body began rejecting the first. Because a donor kidney is foreign to the body, over time the body tries to reject it. In 2009 Barry Freedman was back on dialysis to stay alive.
Watching his uncle suffer prompted Beno Freedman’s path towards stem cell research and the notion that you can reprogram cells for a specific function—turn skin cells into kidney cells, and regenerate an organ that the body won’t reject because, well, it comes from the same body. At Harvard Freedman pursued research on polycystic kidney disease, the most common hereditary type, and wanted to introduce the disease into crafted kidney cells. Know how to create the disease, and you’re close to discovering how to cure it.
For four years, his experiments to create kidneys from stem cells failed, and he scraped by with just enough funding to continue his research. “We didn’t know how to get it to work,” he says. “You’re not sure you can see the light at the end of the tunnel. It begins to wear you down a little bit. And you’re not sure where your career is going to go.” Discouraged, he considered leaving his role as a postdoc at Harvard Medical School altogether.
But Freedman and his three-person lab kept at it—the small, incremental steps to progress through continual trial and error. Lots of errors. Lots of banging their heads up against the wall, Freedman says.
Then one day Freedman saw it. He looked under the microscope, noticed these convoluted, tubular structures, and he knew—those were kidney cells.
Further analysis showed cells, podocytes, that were specific to the kidney and resembled little legs, coming off the body like an octopus, that wrap around blood vessels. Not long after, the mini kidneys on the petri dish started to swell and show signs of polycystic kidney disease. But much about how this was happening remained a mystery.
He left Harvard in 2015 to build his lab at UW, where he learned of the Allen Institute’s edited cells—and where he began using those gene-edited cells. He now employs eight of the institute’s crafted cell lines, or types of cells, in experiments.
Not only are they a great tool for his research, using cells that have proteins tagged with little flashlights (the bioluminescence), but they’re affordable: The Allen Institute’s philosophy lies in “democratizing” science by making them readily available to the public before anything gets published. Scientists all over the world can obtain one of its cell lines for about $600 (essentially the shipping cost) through a third-party distributor, a good thing for gene-editing research labs in the U.S. that compete for federal grants, often at the brink of running out of funding and unsure of what the next few years might bring.
“If you don’t have a way of supporting the research, then it all can stop at any moment,” Freedman says. “There’s actually not nearly enough government money out there to support all the people who want to do cool stuff. Our government money is being spent in many other ways. Build warships, but nobody’s thinking about the war on disease.”
With the Allen Institute’s tags, Freedman sees exactly when and where the cysts form as his mini kidneys develop, connecting those changes to precise structures of the cell. He believes a cure could be on the horizon. “It’s like magic. If we understand how the trick is performed, then we can perform the trick.”

University of Washington researcher Beno Freedman employs gene-edited cells to study possible solutions for polycystic kidney disease.
Image: Ian Bates
At a natural medicine clinic in the University District, Julie Negrin sits in a red leather chair while a mixture of IV fluids makes its way through the tubing and pumps into her arm. Her naturopathic doctor suggested the fluids—a cocktail mixed with 20 different vitamins—as a way to treat her chronic dehydration.
She can’t imagine having just one cancer, she says. Every aspect of her life is a struggle. Her diet has shrunk over time, and in the last two months, she’s had no appetite. But the people in her life keep her fighting, especially her eight nieces and nephews. She loves being an aunt, often referring to them as her kids.
“I just don’t think I’m supposed to go,” she says. She starts to close her eyes as her body relaxes from the vitamin intake. “I see my friends, or see my kids, and I just can’t let go.… I just don’t want to leave those babies.” As soon as the bag empties, an hour into the session, she springs up.
“’Kay, let’s go for a run!” she says, reenergized from the fluids. “Kidding. Okay dear, let’s boogie.”
She puts on her sun hat and grabs her jar of lemonade. She’s on Medicare but pays some money out of pocket today for the IV session.
Since November, she’s had a PICC line—a peripherally inserted central catheter, or a tube inserted through her right arm that connects to a vein near her heart. At home, in a fridge downstairs, she keeps more bags of IV fluids, which she can now administer to herself. A bandage maintains the tube wrapped around her arm at all times, more discreet than other options she once took. It’s less of an inconvenience, she says, though it’s still challenging for her to shower.
If another scientist like Beno Freedman could introduce Lynch syndrome into a cell, he or she could also learn to reverse it. Learn to reverse it, and there could be some kind of treatment or cure—some way to save people like Negrin—whether through an injection or surgery.
Of course, researchers are a long way out from curing hereditary diseases. And when they do get there, they could also discover risks—like, maybe, trying to delete a targeted gene and missing the mark; some clinical trials showed that CRISPR-Cas9 actually introduced mutations into other parts of genes when it deleted the problematic gene elsewhere. It’s precise, sure, but not precise enough. Not yet. Others worry about unwanted side effects. There’s still a lot we don’t know.
Dr. Bill Grady, one of Negrin’s doctors at the Seattle Cancer Care Alliance, says that while there’s a lot of excitement around gene editing now, we’ve been here before. Back in the early 1980s, during his time in medical school, Grady thought that gene therapy—a similar treatment that injects stem cells to regenerate damaged genes—would be moving into clinical trials by 1985.
“And where are we with gene therapy? We’re essentially nowhere,” Grady says. “If [CRISPR-Cas9] fulfills its promise, it could be such a great way to take care of people with Lynch syndrome. But right now, it’s not close enough, unfortunately.”
No, the soonest he expects the technology to help Lynch syndrome patients is 40, maybe 50 years hence. Certainly not in his lifetime, he says—but maybe in our children’s lifetime. Doctors still know little about Lynch syndrome; experts on Lynch have been difficult to find, especially back when Negrin says she first asked about it 10 years before she had been formally diagnosed. And Grady says just one in 10 people who have it know so. Unless you’re aware the syndrome is in your family history, the only way you might know is when you've already developed a tumor. Then test the tumor.
But once doctors establish a family connection with Lynch syndrome, those who potentially have the disease can take a blood test. Negrin’s brother, Rick, says he’ll soon want his 16-year-old son to get tested for the mutation—and the thought that the younger generation could be affected by the same problems Negrin faces gives her all the more motivation to find out as much information as she can.
She’s optimistic. With the way the technology is progressing, Negrin says she has faith the science can catch up in time to help her nieces and nephews. She’d do anything, she says, to help prevent her disease from devastating lives the way it has affected hers.
“This means that my babies might be safe,” Negrin says. “These kids deserve to have a long and full, happy fucking life.”
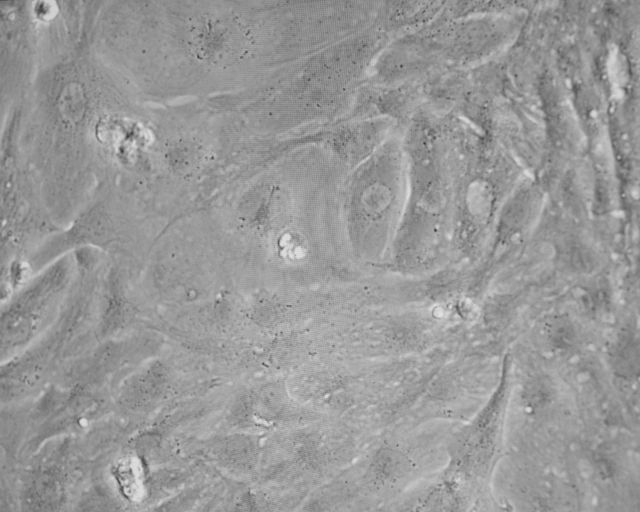
The black-and-white image of cells on a petri dish can get clearer under a fluorescent microscope, which lights up tissue that's tagged with the Cas9 enzyme.
Image: Courtesy the Allen Institute
In the scientific community, discussions on what CRISPR can do often steer into concerns about access and eugenics. Target a disease, and you can target an eye color, a skin color. And when there is a cure, how can we ensure that cure is affordable—not a procedure exclusive to the wealthy?
“The diversity that defines this all is really important in our adaptability and for creating the society that we’re used to,” the Allen Institute’s Rick Horwitz says. “If everyone were equally smart… and equally athletic, I mean, is that where we want to live? I don’t know.”
But, as Horwitz says, you can’t not be excited about the possibilities.
“Nothing is impossible,” Horwitz says. The dreams we once had for science and never thought would come to fruition, he says, are starting to come true.
Updated 10:57am on July 18, 2018, to reflect that stem cells, not heart cells, are shipped to Seattle; that bioluminescence is attached to the template DNA, not Cas9 enzyme; and that CRISPR-Cas9 slices the gene, not cell.




